PRESIDE is the first evidence that docetaxel might reduce enzalutamide resistance when given together. It also suggests that triplet therapy may be helpful in men who have already developed castration resistance.
CAUTION: WHAT YOU DISCLOSE ONLINE ABOUT YOUR HEALTH IS NOT PROTECTED BY HIPAA REGULATIONS.
HIPAA rules bar "covered entities" such as medical practices and hospitals from disclosing our protected health information without first receiving our consent. But for data generated outside the covered entities, there aren't any HIPA protections. Without HIPA protection, we are primarily on our own concerning understanding how companies utilize our personal and health data when we are on social media.
Bipolar Androgen Therapy (BAT): A Patient's Guide
Obtaining Mental Health Services When You Have Cancer
At Cancer ABCs, we understand that cancer diagnosis and treatment are emotionally draining for all. Depression and anxiety are more prevalent in the cancer community, which often impacts treatment outcomes. Depression, anxiety, and fear can make it difficult for patients to participate in therapy, adhere to treatment regimens, and even participate in life. Patients facing mental health issues may have difficulty maintaining a healthy lifestyle, impacting how well cancer therapies work.
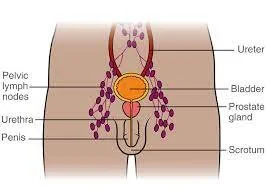
Differential Treatment Response for Hormone Sensitive Men with Nodal Metastases – Nodal Burden as a Prognostic Biomarker
Medicare Open Enrollment Is Approaching
Metformin Did Not Prevent Anthropometric and Metabolic Complications of ADT in Men With Prostate Cancer Receiving Radical Radiotherapy
Pluvicto is Approved By Health Canada
Canada has approved Pluvicto (lutetium (177Lu) vipivotide tetraxetan injection) for the treatment of men with prostate-specific membrane antigen (PSMA) positive metastatic castration-resistant prostate cancer (mCRPC) who have received at least one androgen receptor pathway inhibitor (ARPI) and taxane-based chemotherapy.
Which is Better, 177Lu-PSMA-617 (Pluvicto) or Xofigo (Radium-223)
Several men have asked us if they are better off having 177Lu-PSMA-617 (Pluvicto) or Xofigo, a radiopharmaceutical drug that treats bone metastases. There is no simple answer, and the answer is not the same for each of us. Making a sound decision can be helped by understanding the mode of action and the limits of each treatment.